 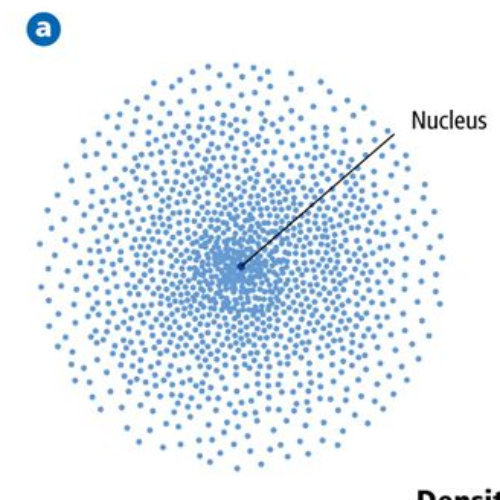
The model does not contemplate the stability of the nucleus, it only refers to explaining the quantum mechanics associated with the movement of electrons within the atom. These atomic orbitals have different levels and sub-levels of energy, and can be defined between electron clouds. The orbitals describe a movement of translation around the nucleus of the atom. These areas of probability are called atomic orbitals. It only establishes a probability zone to locate the electron. This model does not predict the location of the electron, nor does it describe the route it makes within the atom. The electrons move constantly, that is, they do not have a fixed or defined position within the atom. Describes the movement of electrons as standing waves. Orbital 1s, 2s and 2p within a sodium atom. 2.1 Young's experiment: the first demonstration of wave-particle dualityĬharacteristics of the Schrödinger atomic model.1 Characteristics of the Schrödinger atomic model. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
